- info@careermakers.edu.np
- +977 1 4423870
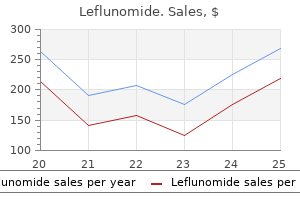
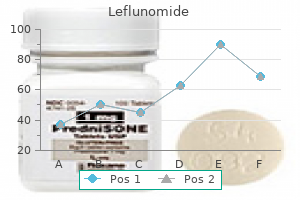
Leflunomide
David Chiu, M.D.
- Assistant Professor of Neurology
- Baylor College of Medicine
- The Methodist Hospital
- Houston, TX
It is the most common cause of a large pleural effusion in the newborn; the etiology is frequently not ascertained (idiopathic) symptoms 9 days post ovulation leflunomide 20 mg order with amex. There are extensive lymphatic channels throughout the body that grow along with the venous system medications known to cause hair loss purchase line leflunomide. The lower extremity and abdominal lymphatics drain into the thoracic duct treatment episode data set discount 20 mg leflunomide fast delivery, which connects into the left innominate vein symptoms 2 weeks after conception purchase cheap leflunomide line. In the chest symptoms 0f kidney stones 10 mg leflunomide order otc, in addition to the thoracic duct, there are bilateral internal mammary lymphatic chains as well as bilateral pleural and lung to bronchomediastinal lymphatic pathways that usually drain into ipsilateral innominate veins. Absence, malformation, disruption, obstruction, or lack of normal venous drainage of the lymphatics may result in abnormal dilated lymphatic channels with diffuse or focal lymphatic malformation masses or free lymphatic fluid including lymphedema, chylothorax, chylopericardium, and chylous ascites. The combination of a soft tissue mass and/or chylothorax with bone and splenic cystic changes is very suggestive of lymphangiomatosis on imaging. Pathologically lymphangioma and lymphangiomatosis are defined as benign neoplasms with proliferating lymphatics. Lymphangioleiomyomatosis is a benign smooth muscle proliferation obstructing veins, lymphatics, and bronchioles, seen in conjunction with tuberous sclerosis, mostly found in young adult women, with innumerable tiny thin-walled cysts within the lungs. Contrast is necessary to sort out fluid versus mass versus atelectatic lung and hemangioma from lymphangioma. Lymphatic lesions do not usually enhance after intravenous contrast except for the septations of macrocystic lesions or when they occur in conjunction with a vascular malformation. Lymphangiography following webspace injection of dye followed by cannulation of a lymphatic channel and injection of Lipiodol to opacify the lymphatics has been performed for many years but is technically challenging, especially in children. Once the thoracic duct is opacified it can be cannulated percutaneously and contrast or other agents injected into the lymphatic system for diagnostic or therapeutic purposes. Nuclear lymphoscintigraphy has been helpful in some select instances of congenital or acquired lymphatic abnormality. The agent technetium 99m albumin is injected subcutaneously, usually in several webspaces of the feet and/or hands, with serial imaging attempting to trace lymphatic drainage pathways. Anatomic definition is somewhat poor using this technique especially as the agent dissipates higher up in the abdomen or chest. Traumatic or iatrogenic entities, idiopathic chylothorax, and lymphangiectasia are often treated conservatively with hyperalimentation and/or a low fat, high protein, medium chain triglyceride diet to decrease lymph flow. More insidious or prolonged chylothorax may be managed by surgical, or more recently percutaneous ligation or ablation of the thoracic duct or other lymphatic ducts. If a discrete malformation is present, management may consist of percutaneous sclerosis (for superficial lesions) or surgical removal. More extensive lesions or those in critical locations may not be able to be surgically removed or sclerosed and may be treated by a variety of palliative methods including various drugs, pleurodesis, and percutaneous ablation to prevent reaccumulation of chylothorax. The more extensive or complex lesions have a tendency to gradually worsen with high long-term morbidity and mortality. Differential diagnosis the differential diagnosis of an opacified hemithorax is quite broad including lung consolidation or atelectasis; pleural effusion; pulmonary, pleural, or even mediastinal mass. The presence of contralateral mediastinal shift implies a spaceoccupying lesion such as a large pleural effusion or an underlying mass or both. Pleural fluid may be simple and transudative (congestive heart failure, accompanying some infections, sympathetic effusion. Decubitus radiographs are not useful when there is marked or complete opacification of a hemithorax since the movement of fluid and visualization of the adjacent lung cannot be seen. The combination of clinical information and multimodality imaging may be needed to help evaluate the most likely diagnostic possibilities. Lymphatic abnormalities of the chest in children include lymphatic dysplasia, lymphangiectasia, lymphatic malformation (lymphangioma), and lymphangiomatosis; these entities overlap. While these abnormalities are rare, their recognition and differentiation from other pathologic entities is important. The observant, knowledgeable radiologist may be the first person to consider lymphatic abnormality as the underlying diagnosis. As the chest, abdomen, soft tissues, and many different organs (especially spleen and bone) may be involved, careful and comprehensive evaluation is required. Percutaneous thoracic duct embolization as a treatment for intrathoracic chyle leaks in infants. Diffuse lymphangiomatosis with genital involvement: evaluation with magnetic resonance lymphangiography. Feasibility of ultrasound-guided intranodal lymphangiogram for thoracic duct embolization. A small piece of echogenic partially atelectatic lung is seen in the center (arrow). A two-year-old girl with congenital pulmonary venous stenosis with continued shortness of breath post multiple pulmonary vein stents. The left upper pulmonary vein was stenotic centrally (arrowhead) and the right upper vein completely thrombosed. Both studies show a large right multicystic chest wall mass extending into the right neck, upper pleura, and mediastinum. Plain chest radiographs are still usually the first imaging studies obtained postnatally in infants with suspected or known congenital heart disease, albeit followed quickly by cardiac echocardiography. While chest radiographs can be entirely normal in infants with significant congenital heart disease or depict only non-specific abnormality, there are some radiographic findings that suggest the presence and nature of the underlying cardiac anomaly. They may also arise from the upper extremity arteries, aorta below the diaphragm, ductus arteriosus, or coronary artery. When systemic collaterals provide blood supply to the lungs, the appearance of the pulmonary vasculature tends to be atypical on imaging studies. Because of relatively poor renal function in the first week of postnatal life, contrast administration is generally avoided unless absolutely necessary. As with all other imaging studies in children, attention should be paid to obtaining a diagnostically useful study at the lowest possible dose. Prostaglandin therapy to maintain patency of the ductus arteriosus had been initiated in the delivery room because of the prenatal echo findings. This was the only systemic arterial supply to the lungs, no additional aorticopulmonary or other collaterals were identified. Additional associated anomalies may include atrial septal defect and common atrioventricular canal (especially in Down syndrome). Tetralogy of Fallot with pulmonary atresia may also be asymptomatic when pulmonary and systemic blood flows are well balanced. Patients generally do well postoperatively with the major problems being residual central or peripheral pulmonary stenoses and pulmonary regurgitation that may lead to subsequent right ventricular dysfunction. Reoperation at a later age to place a homograft or other pulmonary valve is frequently required. The timing and type of surgical intervention depends on the nature and source of pulmonary blood supply. When the ductus arteriosus is the major or sole collateral supply to the lungs (duct dependent) the infant may become critically ill when the ductus begins to close and prostaglandin infusion is needed to keep the duct open. This tends to be a precarious situation and early surgical intervention (a central shunt or complete repair) is usually necessary to ensure a reliable pulmonary blood supply. A large collateral from the coronary circulation may be another indication for early surgery to prevent coronary steal and ischemia. If there are adequate collateral vessels, surgical intervention may be postponed until after the immediate neonatal period. A common pitfall is the false appearance resembling an uplifted cardiac apex in a normal but lordotically positioned chest radiograph. There is marked overinflation of the right lung with leftward cardiomediastinal shift and the suggestion of a right perihilar mediastinal mass. Imaging description A six-month-old girl was evaluated by her primary care physician for noisy breathing while feeding and failure to thrive. The trachea is usually normal in type 1 although a right tracheal bronchus may be present (subtype 1B). Appropriate management addressing both the airway and vascular anomalies is essential for a good outcome. Differential diagnosis Recognition of narrowing, compression, displacement, or poor definition of the airway with or without air trapping on plain chest radiographs raises the question of an underlying lesion. Relatively recent development of tracheoplasty techniques has markedly changed the previously poor prognosis for long segment airway stenosis. Rings, slings, and other things: vascular compression of the infant trachea updated from the midcentury to the millennium  the legacy of Robert E. The evolution of the pulmonary arterial sling syndrome, with particular reference to the need for reoperations because of untreated tracheal stenosis. The configuration suggests congenital tracheal stenosis with complete cartilagenous rings  this was confirmed at bronchoscopy. The presence and severity of symptoms vary with the degree of tracheal compression and a loose ring may be asymptomatic. These vascular anomalies encircle the trachea and esophagus and may occasionally produce dysphagia rather than respiratory symptoms, especially in older children. The most symptomatic vascular ring in infancy is a double aortic arch (persistence of early fetal anatomy) where the ascending and descending aorta and bilateral arches that give rise to ipsilateral separate carotid and subclavian arteries tightly encircle the airway and esophagus. In this situation there is a short residual ligamentous connection between the patent left arch and the diverticular remnant of the distal left arch (Kommerell diverticulum) connected to the descending aorta. Right aortic arch with aberrant left subclavian artery is the most commonly occurring type of vascular ring but tends to be a relatively loose ring and may be asymptomatic. There are other rings that occur uncommonly including left arch with aberrant right subclavian and right Kommerell diverticulum and ductal ligament as well as occasional vascular rings associated with a mirror image right arch, left Kommerell diverticulum, and ductal ligament. Much more commonly a mirror image right arch is associated with congenital heart diseases such as tetralogy of Fallot and truncus arteriosus without a ring. The surgical approach to vascular rings varies with the exact anatomy and is generally intended to break the encircling ring of vessels and relieve tracheoesophageal compression. This most often entails a left thoracotomy with division of the smaller left arch and ligamentum arteriosum (double aortic arch) or just the ligamentum (right arch with aberrant left subclavian). Her mother offered a history of noisy breathing since birth and repeated episodes of wheezing and respiratory infection. The chest radiographs were interpreted as normal in the emergency room; the infant was admitted to the hospital for probable viral upper respiratory infection. The vascular ring encircled the trachea and esophagus with marked compression of the lower trachea at the site of the vascular abnormality. Importance It is essential to include careful evaluation of the airway in the overall assessment of plain chest radiographs. The presence of a right-side aortic arch (often best appreciated by leftward deviation of the airway at the level of the arch) and anterior bowing and narrowing of the lower trachea on the lateral view are the most frequent radiographic clues suggesting the presence of a vascular ring. Although an esophagram was commonly ordered in the past to assess for the possibility of a vascular ring, it is now infrequently obtained for this indication since the images can suggest the presence of a vascular ring but are non-specific as to the exact anatomy. Teaching point An underlying vascular abnormality such as a vascular ring may be responsible for acute or recurrent respiratory symptoms. Careful scrutiny of plain chest radiographs may suggest a possible underlying abnormality that can be further defined by detailed cross-sectional angiographic studies. The major plain radiographic findings that suggest a possible vascular ring are leftward deviation of the trachea by a right-sided aortic arch on the frontal radiograph and anterior tracheal bowing on the lateral radiograph. Rings, slings, and other things: Vascular compression of the infant trachea updated from the mid century to the millennium  the legacy of Robert E. Ultra fast computed tomography and magnetic resonance angiography in pediatric cardiology. Differential diagnosis Differential considerations include other vascular abnormalities that can compress the airway including pulmonary sling and innominate artery syndrome as well as abnormally positioned or enlarged vessels (such as a malpositioned aortic arch in meso- or dextrocardia and right pulmonary hypoplasia or agenesis as well as an enlarged aorta in congenital anomalies such as tetralogy of Fallot or truncus arteriosus). Middle mediastinal masses such as benign or malignant adenopathy, foregut duplication cyst, and esophageal lesions can also produce chronic airway compressive symptoms. Large anterior or posterior mediastinal masses such as lymphoma or neuroblastoma can also displace and compress the airway. Intrinsic airway abnormalities such as malacia, stenosis, bronchospasm, and endobronchial masses may also enter into the differential diagnosis. A 16-month-old girl with double aortic arch vascular ring and recurrent respiratory distress. Note the good definition of the upper trachea with loss of definition of the lower trachea. There is a suggestion of tracheal deviation to the left at this level (arrow), suggesting a right-sided aortic arch. The right arch is larger than the left and there is a markedly narrowed and anteriorly displaced airway encircled by the vessels. Note the larger right arch connecting posteriorly to a distal remnant of the dorsal left arch (diverticulum of Kommerell) (arrow). The left arch is atretic posteriorly between the diverticulum and remaining left arch. The branching pattern is typical, with separate ipsilateral carotid and subclavian arteries arising from each arch. A two and a half-year-old girl with a heart murmur and decreased lower extremity pulses. This curved planar reconstruction demonstrates the components of the loose vascular ring encircling the trachea (T) and esophagus (E). The left carotid artery forms the anterior vascular component, with the right arch to the right and circumflex posterior and left sweep of the arch and a small Kommerell diverticulum (dorsal left arch remnant) forming the posterior components of the ring. The ring is relatively open on the left side where it is completed by a ligamentum arteriosum (not visible). The subsequent branches off the arch are the right carotid and right subclavian arteries (arrowheads).
In addition to the metabolic derangements discussed previously treatment solutions order leflunomide 10 mg with mastercard, diabetes causes other chronic complications that are responsible for the high morbidity and mortality rates associated with this disease medications requiring aims testing generic leflunomide 10 mg buy line. Diabetic complications are largely the result of vascular disease affecting both the microvasculature (retinopathy symptoms 4 days after conception cheap 20 mg leflunomide overnight delivery, nephropathy symptoms underactive thyroid leflunomide 10 mg buy line, and some types of neuropathy) and the macrovasculature (coronary artery disease symptoms restless leg syndrome order generic leflunomide from india, peripheral vascular disease). What is the role of heredity versus the environment in each of the two major types of diabetes mellitus? Pathology & Pathogenesis No matter what the origin, all types of diabetes result from a relative deficiency of insulin action. The resulting metabolic derangements depend on the degree of loss of insulin action. Therefore, low insulin activity is capable of suppressing excessive lipolysis and enhancing fat storage. Higher levels of insulin are required to oppose glucagon effects on the liver and block hepatic glucose output. In normal individuals, basal levels of insulin activity are capable of mediating both of these responses, with the liver, in particular, being exquisitely responsive to changes in pancreatic insulin secretion due to its high sensitivity and exposure to elevated levels of insulin in the portal circulation. However, the ability of skeletal muscle to respond to a glucose load with insulin-mediated glucose uptake requires the stimulated secretion of additional insulin from the pancreas. Mild deficiencies in insulin action are, therefore, frequently manifested by an inability of insulin-sensitive tissues (eg, skeletal muscle which is responsible for 85% of postprandial glucose clearance) to clear glucose loads. Such individuals, most commonly type 2 diabetics with residual insulin secretion but increased insulin resistance, will have abnormal oral glucose tolerance test results and/or high nonfasting (postprandial) glucose levels. However, fasting glucose levels remain normal because sufficient insulin action is present to counterbalance the glucagon-mediated hepatic glucose output that maintains them. Interestingly, skeletal tissue remains insulin sensitive in some prediabetic individuals who can present instead with isolated increases in hepatic glucose output and fasting glucose levels. Hyperglycemia - When elevated glucose levels exceed the renal threshold for reabsorption of glucose, glucosuria results. This causes an osmotic diuresis manifested clinically by polyuria, including nocturia. A significant loss of calories can result from glucosuria, because urinary glucose losses can exceed 75 g/d (75 g × 4 kcal/g = 300 kcal/d). The three "polys" of diabetes - polyuria, polydipsia, and polyphagia - are common presenting symptoms in both type 1 and symptomatic type 2 patients. Weight loss can also occur as a result of both dehydration and loss of calories in the urine. In women, glucosuria can lead to an increased incidence of candidal vulvovaginitis. In uncircumcised men, candidal balanitis (a similar infection of the glans penis) can occur. Diabetic ketoacidosis - A profound loss of insulin activity leads not only to increased serum glucose levels because of increased hepatic glucose output and decreased glucose uptake by insulin-sensitive tissues but also to ketogenesis. In the absence of insulin, lipolysis is stimulated, providing fatty acids that are preferentially converted to ketone bodies in the liver by unopposed glucagon action. Typically, profound hyperglycemia and ketosis (diabetic ketoacidosis) occur in type 1 diabetics, individuals who lack endogenous insulin. Severe hyperglycemia with glucose levels reaching an average of 500 mg/dL can occur if compensation for the osmotic diuresis associated with hyperglycemia fails. Initially, when elevated glucose levels cause an increase in osmolality, a shift of water from the intracellular to the extracellular space and increased water intake stimulated by thirst help to maintain intravascular volume. If polyuria continues and these compensatory mechanisms cannot keep pace with fluid losses - particularly decreased intake as a result of the nausea and increased losses resulting from the vomiting that accompany ketoacidosis - the depletion of intravascular volume leads to decreased renal blood flow. Therefore, glucose levels rise acutely owing to increased glucose production stimulated by these hormones and decreased clearance by the kidney, an important source of glucose clearance in the absence of insulin-mediated glucose uptake. Profound cellular dehydration occurs in response to the marked increase in plasma osmolality. Coma occurs when the effective plasma osmolality reaches 330 mOsm/L (normal: 280Ͳ95 mOsm/L). Because urea is freely diffusible across cell membranes, blood urea nitrogen is not used to calculate the effective plasma osmolality as: Effective osmolality = 2[Na+(mEq/L) + K+(mEq/L)] + Glucose (mEq/L) 18 the increase in ketogenesis caused by a severe lack of insulin action results in increased serum levels of ketones and ketonuria. Insulinopenia is also thought to decrease the ability of tissues to use ketones, thus contributing to the maintenance of ketosis. Na+ is lost in addition to water during the osmotic diuresis accompanying diabetic ketoacidosis. Relative amounts of the two major ketone bodies depend on redox state of the hepatocytes. The nitroprusside reaction, used for clinical testing, only detects compounds with ketone moieties (denoted in blue). If insulin is administered in the absence of fluid and electrolyte replacement, water will move from the extracellular space back into the cells with correction of hyperglycemia, leading to vascular collapse. Insulin administration is also required to inhibit further lipolysis, thus eliminating substrates for ketogenesis, and to inhibit hepatic ketogenesis, thereby correcting ketoacidosis. During treatment of diabetic ketoacidosis, measured serum ketones may transiently rise instead of showing a steady decrease. This is an artifact because of the limitations of the nitroprusside test that is often used at the bedside to measure ketones in both serum and urine. With insulin treatment, fatty acid oxidation decreases and the redox potential of the liver shifts back in favor of acetoacetate formation. Therefore, although the absolute amount of hepatic ketone body production is decreasing with treatment of diabetic ketoacidosis, the relative amount of acetoacetate production is increasing, leading to a transient increase in measured serum ketones by the nitroprusside test. These episodes are frequently precipitated by decreased fluid intake such as can occur during an intercurrent illness or in older debilitated patients who lack sufficient access to water and have abnormal renal function hindering the clearance of excessive glucose loads. The mechanisms underlying the development of hyperosmolality and hyperosmolar coma are the same as in diabetic ketoacidosis. However, because only minimal levels of insulin activity are required to suppress lipolysis, these individuals have sufficient insulin to prevent the ketogenesis that results from increased fatty acid flux. Because of the absence of ketoacidosis and its symptoms, patients often present later and, therefore, have more profound hyperglycemia and dehydration; glucose levels often range from 800Ͳ400 mg/dL. Therefore, the effective osmolality exceeds 330 mOsm/L more frequently in these patients than in those presenting with diabetic ketoacidosis, resulting in a higher incidence of coma. Although ketosis is absent, mild ketonuria can be present if the patient has not been eating. Mortality is 10 times higher than in diabetic ketoacidosis because the type 2 diabetics who develop hyperosmolar nonketotic states are older and often have other serious precipitating or complicating illnesses. For example, myocardial infarction can precipitate hyperosmolar states or can result from the alterations in vascular blood flow and other stressors that accompany severe dehydration. However, acidosis, insulinopenia, and elevated glucose levels cause a shift of K+ out of cells, thus maintaining normal or even elevated serum K+ levels until acidosis and hyperglycemia are corrected. With administration of insulin and correction of acidosis, serum K+ falls as K+ moves back into cells. Without treatment, K+ can fall to dangerously low levels, leading to potentially lethal cardiac arrhythmias. Therefore, K+ supplementation is routinely given in the treatment of diabetic ketoacidosis. Similarly, phosphate depletion accompanies diabetic ketoacidosis, although acidosis and insulinopenia can cause serum phosphorus levels to be normal before treatment. Phosphate replacement is provided only in cases of extreme depletion given the risks of phosphate administration. Although serum Na+ levels can be decreased owing to the osmotic effects of glucose, hypertriglyceridemia can interfere with some common procedures used to measure serum Na+. This causes pseudohyponatremia (ie, falsely low serum Na+ values, due to overestimation of actual serum volume). Nausea and vomiting often accompany diabetic ketoacidosis, contributing to further dehydration. Abdominal pain, present in 30% of patients, may be due to gastric stasis and distention. Amylase is frequently elevated (90% of cases), in part because of elevations of salivary amylase, but it is usually not associated with symptoms of pancreatitis. Leukocytosis is frequently present and does not necessarily indicate the presence of infection. Diabetic ketoacidosis is treated by replacement of water and electrolytes (Na+ and K+) and administration of insulin. With fluid and electrolyte replacement, renal perfusion is increased, restoring renal clearance of elevated blood glucose, and counterregulatory hormone production is decreased, thus decreasing hepatic glucose production. Hypoglycemia often occurs during exercise or with fasting, states that normally are characterized by slight elevations in counter-regulatory hormones and depressed insulin levels. Under normal circumstances, low insulin levels in these conditions are permissive for the counter-regulatory hormonemediated mobilization of fuel substrates, increased hepatic glucose output, and inhibition of glucose disposal in insulinsensitive tissues. In addition, the fall in insulin secretion by the pancreatic cell in response to low glucose levels is an important stimulus for increased secretion of glucagon. However, in diabetic patients, all of these responses fail when insulin is maintained at excessive levels (relative to plasma glucose) due to excessive exogenous insulin dosing or endogenous glucose-independent insulin stimulation. The acute response to hypoglycemia is mediated by the counter-regulatory effects of glucagon and catecholamines (Table 18͵). However, the glucagon response can be inadequate in diabetes, increasing the importance of adrenal epinephrine secretion. A characteristic set of symptoms (night sweats, nightmares, morning headaches) also accompanies hypoglycemic episodes that occur during sleep (nocturnal hypoglycemia). Moreover, recent episodes of hypoglycemia reduce the adrenal epinephrine response to subsequent hypoglycemia and cause hypoglycemia unawareness by reducing the sympathoadrenal response and associated neurogenic symptoms via unknown mechanisms. This hypoglycemia-induced autonomic failure, which is distinct from diabetic autonomic neuropathy, is reversed by avoidance of hypoglycemia but exacerbated by exercise or sleep, both of which can similarly decrease the sympathoadrenal response to a given level of hypoglycemia. Acute treatment of hypoglycemia in diabetic individuals consists of rapid oral administration of glucose at the onset of warning symptoms or the administration of exogenous glucagon intramuscularly by others when neuroglycopenic symptoms preclude oral self-treatment. Rebound hyperglycemia can occur after hypoglycemia because of the actions of counter-regulatory hormones (Somogyi phenomenon), an effect that can be aggravated by excessive glucose administration. Chronic Complications Over time, diabetes results in damage and dysfunction in multiple organ systems (Table 18Ͷ). While the importance of glycemic control in influencing the occurrence of microvascular complications is undisputed, genetic factors also clearly play a role. For example, evidence from a variety of studies suggests that approximately 40% of type 1 diabetics are particularly susceptible to the development of severe microvascular complications. The identity of genetic factors associated with microvascular disease risk is the subject of ongoing investigations, which have already identified numerous candidate genes coding for the extracellular matrix, transcription factors, growth factor signaling, and/or erythropoietin. Neuropathy also causes increased morbidity, particularly by virtue of its role in the pathogenesis of foot ulcers. The end result of these changes in the microvasculature is an increase in protein accumulation in vessel walls, endothelial cell dysfunction, loss of endothelial cells, and, ultimately, occlusion. Many cells contain aldose reductase, an enzyme that converts toxic aldehydes to their respective alcohols (polyol pathway). While aldose reductase has a low affinity for glucose, under conditions of intercellular hyperglycemia, this pathway can account for up to one-third of glucose flux, converting glucose to sorbitol. While polyol pathwayÎediated damage appears to be a prominent feature in nerve cells, its role in the vasculature is less clear. In diabetics, elevated glucose leads to increased glycation of HbA within red blood cells. The reversible formation of glycated proteins (Amadori products), such as hemoglobin A1c, through a complex series of chemical reactions, or the direct oxidation of glucose and its metabolites (eg, glyceraldehyde-3 phosphate, G3P), result in the production of reactive dicarbonyls. These outpouchings in the capillary wall are due to loss of surrounding pericytes that support the capillary walls. Fat that has leaked from excessively permeable capillary walls appears as shiny yellow spots with distinct borders (hard exudates) forming a ring around the area of leakage. As retinopathy progresses, signs of ischemia appearing as background retinopathy worsen (preproliferative stage). Occlusion of capillaries and terminal arterioles causes areas of retinal ischemia that appear as hazy yellow areas with indistinct borders (cotton wool spots or soft exudates) because of the accumulation of axonoplasmic debris at areas of infarction. Retinopathy can progress to a second, more severe stage characterized by the proliferation of new vessels (proliferative retinopathy). It is hypothesized that retinal ischemia stimulates the release of growth-promoting factors, resulting in new vessel formation. However, these capillaries are abnormal, and traction between new fibrovascular networks and the vitreous can lead to vitreous hemorrhage or retinal detachment, two potential causes of blindness. Basement membranes of the glomerular capillaries are thickened and can obliterate the vessels; the mesangium surrounding the glomerular vessels is increased owing to the deposition of basement membrane-like material and can encroach on the glomerular vessels; and the afferent and efferent glomerular arteries are also sclerosed. Glomerulosclerosis is usually diffuse but in 50% of cases is associated with nodular sclerosis. This nodular component, called Kimmelstiel-Wilson nodules after the investigators who first described the pathologic changes in diabetic kidneys, is pathognomonic for diabetes but is present in only 30% of patients with microalbuminuria. The hexosamine pathway contributes to insulin resistance, producing substrates that, when covalently linked to transcription factors, stimulate the expression of proteins, such as transforming growth factor and plasminogen activator inhibitor, that enhance microvascular damage. Evidence suggests that all four of these pathways may actually be linked by a common mechanistic element: hyperglycemiainduced oxidative stress. In particular, the increase in electron donors that results from shunting glucose through the tricarboxylic acid cycle increases mitochondrial membrane potential by pumping proteins across the mitochondrial inner membrane. This increased potential prolongs the half-life of superoxide generating enzymes, thus increasing the conversion of O2 to O2Í® Albuminuria is thought to be due to a decrease in the heparan sulfate content of the thickened glomerular capillary basement membrane. Heparan sulfate, a negatively charged proteoglycan, can inhibit the filtration of other negatively charged proteins, such as albumin, through the basement membrane; its loss, therefore, allows for increased albumin filtration. Diabetic nephropathy is defined clinically by the presence of more than 300 mg of urinary protein per day, an amount that can be detected by routine urinalysis. In diabetic nephropathy (unlike other renal diseases), proteinuria continues to increase as renal function decreases. Therefore, end-stage renal disease is preceded by massive, nephrotic-range proteinuria (>4 g/d).
Formation medications requiring prior authorization buy generic leflunomide 20 mg, Secretion symptoms of ms buy 20 mg leflunomide fast delivery, & Metabolism of Catecholamines the adrenal medulla secretes three catecholamines: epinephrine symptoms of breast cancer order leflunomide 20 mg fast delivery, norepinephrine medications 1 gram cheap 20 mg leflunomide overnight delivery, and dopamine treatment plant buy generic leflunomide 10 mg line. Secretion occurs after release of acetylcholine from the preganglionic neurons that innervate the medullary cells. In humans, most (80%) of the catecholamine output of the adrenal medulla is epinephrine. Approximately 70% of the epinephrine and norepinephrine and 95% of the dopamine found in plasma are conjugated to sulfate and inactive. Medullary cells secrete catecholamines after release of acetylcholine from the preganglionic neurons that innervate them. Catecholamine secretion is low in the basal state and is reduced even further during sleep. In emergency situations, there is increased adrenal catecholamine secretion as part of a generalized sympathetic discharge that serves to prepare the individual for stress ("fight-or-flight" response). Physiological stress such as psychological, physical (eg, mechanical, thermal), and metabolic (eg, hypoglycemia, exercise) stress leads to catecholamine secretion. Mechanism of Action of Catecholamines the effects of epinephrine and norepinephrine are mediated by their actions on two classes of receptors: - and -adrenergic receptors (Table 12ͱ). Alpha receptors are subdivided into 1 and 2 receptors and receptors into 1, 2, and 3 receptors. Beta1 receptors mediate an increased rate and force of myocardial contraction and stimulate lipolysis and renin release. Intracellular post-receptor signaling is different for each subclass of adrenergic receptor. Stimulation of 1-adrenergic receptors results in an increase in intracellular Ca2+ concentrations. First, there is activation of phospholipase C by the guanine nucleotide binding stimulatory protein, Gs. Phospholipase C hydrolyzes the membrane-bound phospholipid, phosphatidylinositol-4,5-bisphosphate, to generate two second messengers: diacylglycerol and inositol-1,4,5-trisphosphate. Diacylglycerol in turn activates protein kinase C, which phosphorylates various cellular substrates. Inositol-1,4,5trisphosphate stimulates release of intracellular Ca2+, which then initiates various cellular responses. The mechanism involves receptor interaction with an inhibitory G protein, Gi, leading to inhibition of adenylyl cyclase. The Gi protein also stimulates K+ channels and inhibits voltage-sensitive calcium channels. On the other hand, -adrenergic receptors stimulate adenylyl cyclase through the mediation of Gs. The Gs protein can also directly activate voltage-sensitive Ca2+ channels in the plasma membrane of cardiac and skeletal muscle. Most catecholamine metabolism takes place within the same cells where they are synthesized, mainly because of leakage of catecholamines from vesicular stores into the cytoplasm. These vesicular stores exist in a dynamic equilibrium, with outward passive leakage counterbalanced by inward active transport that is controlled by vesicular monoamine transporters. In catecholaminergic neurons, the presence of monoamine oxidase in the cytoplasm leads to formation of reactive catecholaldehydes. Production of these toxic aldehydes is dependent on the dynamics of the vesicular-axoplasmic monoamine exchange and an enzyme-catalyzed conversion to nontoxic acids or alcohols. In sympathetic nerves, the aldehyde produced from norepinephrine is converted to 3,4-dihydroxyphenylglycol. Compared with intraneuronal deamination, extraneuronal O-methylation of norepinephrine and epinephrine to metanephrines represents minor pathways of metabolism. The 1- and 1-adrenergic receptors are preferentially stimulated by norepinephrine, especially that released by nerve endings. In contrast, the 2and 2-adrenergic receptors are generally situated in postjunctional sites in organs and tissues (eg, uterine and bronchial skeletal muscle) remote from sites of norepinephrine release. The 2- and 2-adrenergic receptors are preferentially stimulated by circulating catecholamines, especially epinephrine. Differences in tissue distribution, accessibility by nerve fibers, preferences for epinephrine versus norepinephrine, and differences in postreceptor signaling are thus responsible for the diverse effects of catecholamines in an organ- and cellspecific manner. Effects of Catecholamines the catecholamines have been termed fight-or-flight hormones because their effects on the heart, blood vessels, smooth muscle, and metabolism assist the organism in responding to stress. In the peripheral circulation, norepinephrine produces vasoconstriction in most organs (via 1 receptors). Epinephrine produces vasodilation via 2 receptors in skeletal muscle and liver and vasoconstriction elsewhere. The rise in blood pressure stimulates the carotid and aortic baroreceptors, resulting in reflex bradycardia and a fall in cardiac output. Epinephrine causes a widening of pulse pressure but does not stimulate the baroreceptors to the same degree, so the pulse rises and cardiac output increases. Hence, pheochromocytomas or other tumors of the adrenal medulla, which usually secrete norepinephrine, lead to vasoconstriction and an increase in blood pressure. The effects of catecholamines on metabolism include effects on glycogenolysis, lipolysis, and insulin secretion, mediated by both - and -adrenergic receptors. These metabolic effects result primarily from the action of epinephrine on four target tissues: liver, muscle, pancreas, and adipose tissue (see Table 12ͱ). The result is an increase in the levels of circulating glucose and free fatty acids. The increased supply of these two substances helps provide an adequate supply of metabolic fuel to the nervous system and muscle during physiologic stress. The amount of circulating plasma epinephrine and norepinephrine needed to produce these various effects has been determined by infusing the catecholamines into resting subjects. For norepinephrine, the threshold for the cardiovascular and metabolic effects is a plasma level of about 1500 pg/ mL, or about five times the basal level. In normal individuals, the plasma norepinephrine level rarely exceeds this threshold. However, for epinephrine, the threshold for tachycardia occurs at a plasma level of about 50 pg/mL, or about twice the basal level. The threshold for increasing systolic blood pressure and lipolysis is at about 75 pg/mL; for increasing glucose and lactate, about 150 pg/mL; and for increasing insulin secretion, about 40 pg/mL. Peripherally, in small doses, injected dopamine produces renal vasodilation, probably by binding to a specific dopaminergic receptor. In moderate doses, it also produces vasodilation of 323 the mesenteric and coronary circulation and vasoconstriction peripherally. It has a positive inotropic effect on the heart, mediated by action on the 1-adrenergic receptors. Moderate to large doses of dopamine increase the systolic blood pressure without affecting diastolic pressure. Overview of Adrenal Medullary Disorders Pheochromocytoma is an uncommon tumor of adrenal medullary tissue that causes production of excessive amounts of catecholamines. Patients typically present with sustained or episodic hypertension or with a syndrome characterized by episodic palpitations, tachycardia, chest pain, headache, anxiety, pallor, excessive sweating and hyperglycemia. Pheochromocytomas are closely related to paragangliomas, which sometimes are termed extra-adrenal pheochromocytomas. Most parasympathetic paragangliomas are found in the head and neck area (eg, carotid body, vagal nerve) and either do not secrete any catecholamines or rarely secrete norepinephrine. Most sympathetic paragangliomas arise in the abdomen and often secrete norepinephrine. What physiologic processes do each subtype of catecholamine receptor control, and how do catecholamines bring about each of these physiologic process? Other tumors of the adrenal medulla or its embryonic precursors include neuroblastomas and ganglioneuromas. In response to therapy (or even spontaneously), neuroblastomas can differentiate into ganglioneuromas. Both of these tumors secrete catecholamines, but symptoms due to catecholamine excess are usually absent because they do not reach the levels observed with pheochromocytomas. Absence of the adrenal medulla (eg, after bilateral adrenalectomy) is usually well tolerated, though sometimes symptoms such as orthostatic hypotension may be observed. These tumors secrete excessive amounts of epinephrine, norepinephrine, or both (rarely dopamine). Most pheochromocytomas secrete norepinephrine and cause sustained or episodic hypertension. Pheochromocytomas that secrete epinephrine cause hypertension less often; more frequently, they produce episodic hyperglycemia, glucosuria, and other metabolic effects. Pheochromocytomas occur in both sexes and in all age groups but are most often diagnosed in the fourth or fifth decade of life. Compared with adults, children with pheochromocytomas are more likely to have multifocal and extra-adrenal tumors, and a causal familial syndrome must always be excluded. The diagnosis is important because sudden release of catecholamines from these tumors during anesthesia, surgery, or obstetric delivery may prove fatal. Pheochromocytoma was classically referred to as "the 10% tumor" because 10% occur in extra-adrenal paraganglia, 10% are outside the abdomen, 10% are multiple, 10% are bilateral, about 10% are not associated with hypertension, 10% occur in children, and 10% are malignant. So, previously, it was thought that about 10% occur as part of a familial syndrome, but now it appears that actually about 20ͳ0% of cases are familial. Also, occurrence at extra-adrenal sites seems to be higher (9Ͳ3%) and multifocal pheochromocytomas can be found in roughly one third of childhood cases. Etiology Several genetic syndromes, all transmitted in an autosomal dominant fashion, are associated with an increased risk of pheochromocytoma and sympathetic or parasympathetic nervous system paragangliomas. The 634 cysteine residue is part of an intramolecular sulfide bridge between associated cysteine residues. Indeed, while a carrier of a maternally inherited allele can propagate the defect to offspring, this mutant allele does not increase the risk for a paraganglioma. Given the high frequency of germline mutations, genetic counseling and genetic testing are recommended for all patients with pheochromocytomas or paragangliomas, particularly those with a positive family history, multifocal disease, or a diagnosis before age 50 years. Genetic testing may also be useful in screening families of carriers of mutations detected. Almost all pheochromocytomas (about 90%) occur in the abdomen, and most of these (85%) are in the adrenal medulla. However, such patients also usually experience paroxysms related to transient increases in catecholamine release. The long-term exposure to high levels of circulating catecholamines seems not to produce the classic hemodynamic responses observed after acute administration of catecholamines. This may be due in part to desensitization of the cardiovascular system to catecholamines and may explain why some patients with pheochromocytomas are entirely asymptomatic. He also had a medullary carcinoma of the thyroid and a large pheochromocytoma in the opposite adrenal. The classical pentad of symptoms in patients with pheochromocytoma consists of: headache, palpitation, perspiration, pallor, and orthostasis. In about half of cases, hypertension is sustained but the blood pressure shows marked fluctuations, with peak pressures during symptomatic paroxysms. During a hypertensive episode, the systolic blood pressure can rise to as high as 300 mm Hg. The increased total peripheral vascular resistance is probably primarily responsible for the maintenance of high arterial pressures. Hypertensive crisis may be precipitated by a variety of drugs, including tricyclic antidepressants, antidopaminergic agents, metoclopramide, and naloxone. Beta blockers should not be administered until alpha blockade has been established. Otherwise, blockade of 2-adrenergic receptors, which promote vasodilation, will allow unopposed -adrenergic receptor activation and produce marked vasoconstriction and hypertension. Peripheral vasoconstriction, mediated by receptors, causes both facial pallor and cool, moist hands and feet. Chronic vasoconstriction of the arterial and venous beds leads to a reduction in plasma volume and predisposes to postural hypotension. In others, orthostatic hypotension is associated with decreased cardiac stroke volume and an impaired response of total peripheral vascular resistance to changes in posture, perhaps indicative of diminished arteriolar and venous responsiveness. The reduced responsiveness of the vasculature to norepinephrine in patients with pheochromocytoma is probably related to downregulation of -adrenergic receptors resulting from persistent elevations of norepinephrine levels. They are highly vascular tumors and frequently have cystic, necrotic, or hemorrhagic areas. Microscopically, the tumor consists of large pleomorphic cells arranged in sheets separated by a highly vascular stroma. In the cytoplasm, there are catecholamine-containing storage granules similar to those in normal adrenal medullary cells. Mitoses are rare, but tumor invasion of the adrenal capsule and blood vessels is common even in benign pheochromocytomas. Malignancy is established only when a metastasis is found in a site where chromaffin cells are not usually demonstrated (eg, liver, lung, bone, or brain). Pathogenesis Most pheochromocytomas release predominantly norepinephrine, but most also release epinephrine (Table 12ʹ). Rarely, a pheochromocytoma releases mostly or only epinephrine and very rarely mostly or only dopamine. In about half of patients with pheochromocytoma, clinical manifestations vary in intensity and occur in an episodic or paroxysmal fashion. The sudden catecholamine excess causes hypertension, palpitations, tachycardia, chest pain, headache, anxiety, blanching, and excessive sweating.
Unfortunately treatment kidney stones order leflunomide 10 mg online, for several reasons symptoms 7 days after ovulation generic leflunomide 20 mg without a prescription, they often have high mortality rates and a high incidence of metastases when they are diagnosed symptoms quitting smoking order 10 mg leflunomide amex. Some organs are located deep and are relatively inaccessible to palpation (ovaries) medications zanaflex buy leflunomide 20 mg low price. Others have few sensory nerves (ovary abro oil treatment order genuine leflunomide line, fallopian tubes) and hence remain asymptomatic. Additionally, the breasts have large amounts of adipose tissue, which can make early detection of breast cancer difficult. Disorders of the female reproductive system can also occur as a result of disease in other organs whose function affects reproductive organs (eg, the brain, hypothalamus, pituitary, thyroid, adrenals, kidney, and liver). Conversely, disorders of the reproductive system can cause disorders in other tissues. Ovarian hormones are necessary for the maintenance and health of most tissues in women. Alterations in these hormones can lead to osteoporosis (loss of bone mass), atrophy and inflammation of estrogen-deprived tissues (eg, atrophic vaginitis), atherogenesis and alterations in cardiovascular compliance, and an increased risk of some forms of cancer (eg, endometrial carcinoma as a consequence of estrogen excess and progesterone deficiency). Dysfunction of the reproductive system also can contribute to unique variants of systemic disorders, such as gestational diabetes and the hypertensive syndrome of preeclampsia-eclampsia. How do female reproductive system disorders present during the reproductive years? To what might you ascribe the lack of reduction in mortality rate from ovarian cancer in contrast to cervical cancer? The two ovaries contain thousands of follicles, each with an oocyte surrounded by a layer of granulosa cells and thecal cells. These supporting cells produce steroids and paracrine products important in follicular maturation and coordination of events in reproduction. The fallopian tubes, which are open to the peritoneal space, connect the ovaries to the uterus. The uterus contains an internal hormone-sensitive mucosal lining, the endometrium. The outlines of the lobules do not exist in vivo but are shown for instructional purposes. Surrounding the endometrium is the smooth muscle layer of the uterus, the myometrium. Contractions of the myometrium lead to menstrual cramps or expel the fetus at parturition. The cervix is contiguous with the uterus and is the conduit for passage of menses or the fetus into the vagina, the muscular tube opening into the vulva. Once at the genital ridge, they multiply and induce male or female gonads depending on the identity of the sex chromosomes. Until week 8 of gestation, the sex of the embryo cannot be determined morphologically; therefore, this period is termed the indifferent phase of sexual development. After this time, differentiation of the internal and external genitalia occurs, determining the phenotypic sex of the individual, which becomes fully developed after puberty. During embryogenesis, the internal genitalia are formed from a dual genital duct system within the urogenital ridge. The first to form is the wolffian duct, followed by the m𬬥rian duct, which is dependent on prior wolffian duct development. After 8 weeks of gestation, production of anti-m𬬥rian hormone by Sertoli cells in the fetal testes leads to regression of the m𬬥rian ducts, whereas production of testosterone by the Leydig cells leads to persistence of the wolffian duct and subsequent development of the prostate, epididymis, and seminal vesicles. In the absence of these secretions, female internal reproductive organs are formed from the m𬬥rian ducts, and the wolffian structures degenerate. Similarly, the external genitalia of males develop in the presence of dihydrotestosterone; in the absence of this hormone, the common embryologic structures give rise to female external genitalia. During female development, the female ovaries contain about 7 million oogonia by 24 weeks of gestation. The majority of these cells die during intrauterine life, leaving only about 1 million primary oocytes at birth. Completion of the first meiotic division does not occur until the time of ovulation, and the second meiosis is completed with fertilization. These changes allow estrogendependent tissues, such as the breasts and the endometrium, to begin their maturation. The appearance of breast development is referred to as thelarche, and the first menstrual period is termed menarche. What is the difference between the chromosomal, gonadal, and phenotypic sex of an individual? Approximately what percentage of the total number of oocytes present in the ovaries of a female at birth completes their maturation and is released upon ovulation over the course of her reproductive life? Before about age 10 years in girls, gonadotropin secretion is at low levels and does not display a pulsatile character. With this coordination, cyclic changes during the course of the menstrual cycle allow the reproductive organs to perform specific functions at different points in time to optimize the chances for successful reproduction. An atretic follicle is shown in the center, and the structure of the wall of the mature follicle is detailed at the upper right. The follicular phase typically lasts 12-14 days and culminates in the production of a mature oocyte. The follicular phase is followed by ovulation, in which the dominant follicle releases its mature oocyte to be transported through the uterine tubes for fertilization and subsequent implantation in a receptive uterus. The third, luteal, phase also averages 14 days and is characterized by luteinization of the ruptured follicle to produce the corpus luteum. The neuroendocrine axis involves the brain, hypothalamus, pituitary, and the ovary. This pulsatility is required for proper activation of its receptor located on the gonadotropes, which are cells located in the anterior pituitary. After this surge, high levels of progesterone produced by the corpus luteum suppress gonadotropin release for the duration of the luteal phase. Non-oral formulations also have been developed, including long-term intrauterine and subdermal systems that deliver progestins. A transdermal patch allows absorption of estrogen and progestin without "first-pass" metabolism in the liver. Transvaginal absorption is also available with a soft ring placed monthly in the vagina. Each of these formulations provides contraceptive efficacy equal to or better than oral contraceptive pills. Androgens diffuse into the granulosa cells to be converted to estrogens through the enzymatic reaction of aromatization. Changes in prostaglandins and proteases allow digestion of the follicular wall leading to oocyte extrusion and ovulation. The follicular cells remaining after ovulation develop into a structure called the corpus luteum, which synthesizes and releases large amounts of both estradiol and progesterone. The uterine compartment reacts to the steroids produced from the ovaries throughout the menstrual cycle. During the follicular phase, the endometrium proliferates under the influence of estrogen, creating straight glands with thin secretions and microvascular proliferation. Additionally, the endometrium secretes a number of endocrine and paracrine factors (Table 22ͱ). In the absence of pregnancy, the corpus luteum cannot sustain the high levels of progesterone production and the endometrial vasculature cannot be maintained. The ovary secretes three types of steroids: progesterone, containing 21 carbons; androgens, containing 19 carbons; and estrogens, containing 18 carbons. Steroid synthesis occurs by conversion from cholesterol in a series of oxidative biochemical reactions catalyzed by enzymes in the mitochondria and the endoplasmic reticulum (see Chapter 21). This steroid is further modified in the endoplasmic reticulum to generate the various steroid hormones. Because steroids are synthesized by a cascade of enzyme reactions in various pathways, a block in one step (eg, resulting from a congenital enzyme defect or inhibition by certain drugs) can result in lack of synthesis of downstream products and "spillover" of precursors. Such defects are the hallmark of congenital adrenal hyperplasia (discussed in Chapter 21). In this way, the pattern of gene expression is changed in the various steroid-responsive tissues (ie, those that contain steroid receptors). Membrane-bound steroid receptors also have been shown to activate phosphorylation cascades typically regulated by growth factors. Contraception Birth control pills are a pharmacologic means of preventing pregnancy by disrupting the precise timing of hormone-directed events necessary for reproduction. Current formulations include progestins alone as well as combinations of estrogens and progestins. However, other contraceptive actions include effects on estrogen- and progesterone-sensitive tissues, such as inducing antifertility changes in cervical mucus and the endometrial lining that are unfavorable to sperm transport and embryonic implantation, respectively. How does the structure of the uterine lining differ in the midfollicular versus the midluteal stages, and for what reproduction-related events is each stage optimized? What products are made by a granulosa cell in the dominant follicle over the course of its lifetime? Fertilization requires successful ovulation, capture of the mature oocyte by the fimbria of the fallopian tubes, and transport of the zygote to the uterus. Because fertilization usually occurs in the ampulla, it also requires effective transport of viable sperm into the distal tube. The placenta allows intimate apposition of maternal and fetal circulations for exchange of nutrients, oxygen, and waste products. This reflects the action of a special zone in the fetal adrenal cortex engaged in androgen production. This switch may play a role in triggering the onset of labor by modulating the expression of progesterone receptors in the myometrium. In addition to the changes in organs with pregnancy-specific functions, physiologic changes occur in essentially every maternal organ system. These include increased blood volume (increased by more than 40% by the middle of the third trimester), increased total body water (increased by 6͸ L), and increased cardiac output because of increased stroke volume (increased by 30%) and heart rate (increased by 15%). A striking increase in minute ventilation (increased by 50% compared with nonpregnant state) without any change in respiratory rate is observed as a result of increased tidal volume (Chapter 9). Dramatic increases in renal blood flow and glomerular filtration rate (increased by 40%) are also seen. Most of these alterations are related in complex ways to the effects of hormones produced in pregnancy. Effects of Ovarian Steroids in Pregnancy the physiological effects of various sex steroids in pregnancy are incompletely understood. The demonstrated and proposed roles of progesterone in pregnancy include (1) promotion of implantation; (2) suppression of the maternal immune response to fetal antigens, thus preventing rejection of the allogeneic fetus; (3) increased cardiovascular compliance; (4) provision of substrate for manufacture by the fetal adrenal of glucocorticoids and mineralocorticoids; (5) maintenance of myometrial quiescence through gestation; and (6) a role in parturition. Estrogens contribute to (1) volume expansion; (2) cardiac remodeling; and (3) preparative production of clotting factors, anticipating blood loss that commonly follows delivery. This "counterregulatory" hormone (ie, a hormone whose actions oppose those of insulin) appears to serve as a defense against fetal hypoglycemia. From a metabolic standpoint, pregnancy is a form of "accelerated starvation" characterized by fasting hypoglycemia, as fuel substrates produced by the mother are consumed by the growing fetus. However, in the event of glucose deprivation, ketones provide a ready emergency fuel supply (as they do in starvation) for both the mother and, via the placenta, for the fetus. During puberty, rising estrogen levels stimulate breast growth as one of a number of female secondary sexual characteristics. Breast growth involves both proliferation and branching of lactiferous ducts as well as accumulation of adipose and connective tissue. In the mature breast, each terminal lactiferous duct drains clusters of tubuloalveolar secretory units lined by milk-secreting epithelial cells and is suspended in connective and adipose tissue well populated with lymphocytes. Both the pubertal and pregnant phases of breast growth require the permissive influence of glucocorticoids, thyroxine, and insulin for full development, and their actions are potentiated by estrogen and progesterone. There may also be effects of prolactin directly on the ovary that contribute to lactational anovulation and amenorrhea. However, it should be noted that the contraceptive effect of prolactin is only moderate and, therefore, of low reliability. Ten years before menopause, at approximately age 40 years, reproductive function starts to diminish. This is manifested as a decreased frequency of ovulation and alterations in menstrual patterns. Higher levels of estradiol circulate, particularly in the follicular phase from ages 35ʹ8 years, and then estradiol levels sharply decline just prior to menopause. This transitory period of diminishing reproductive function approaching menopause is termed the climacteric. During the climacteric transition, the hormonal status of women changes from a cyclic high-estrogen state to a steadystate low-estrogen postmenopausal state. This leads to vasomotor symptoms such as hot flushes ("hot flashes"), sweating, and chills. Psychologic symptoms such as irritability, tension, anxiety, and depression may also be observed. In addition to atrophy of estrogen-dependent tissues such as the vaginal epithelium, a gradual loss in bone density leading to osteoporosis can occur. A modest degree of androgen production from thecal cells of the residual ovarian stroma continues even in the absence of follicular growth. The significance of peripheral aromatization in relation to severity of symptoms of menopause varies in different individuals. In the medical literature, menopause was often viewed as an "endocrinopathy," specifically as a disorder of estrogen deficiency. Actual lactation, or milk release, however, is inhibited by the high levels of placental steroids present before birth. After delivery of the placenta, estrogen and progesterone levels fall dramatically, removing this block.
Generic 10 mg leflunomide fast delivery. Aids What Do We Know Now - Malayalam | Hinduism.
References
- Sakr, W.A., Haas, G.P., Cassin, B.F., Pontes, J.E., Crissman, J.D. The frequency of carcinoma and intraepithelial neoplasia of the prostate in young male patients. J Urol 1993; 150:379-385.
- Valerio M, Dickinson L, Ali A, et al: Nanoknife electroporation ablation trial: a prospective development study investigating focal irreversible electroporation for localized prostate cancer, J Urol 197(3 Pt 1):647n654, 2017.
- Steinberg MJ, Herrera AF. Management of parotid duct injuries. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2005;99:136-141.
- Fox KA, Steg PG, Eagle KA, et al: Decline in rates of death and heart failure in acute coronary syndromes, 1999-2006.
- Kradin R. Pulmonary immune response. In Kradin R, Robinson B, eds. Immunopathology of the Lung. Boston: Butterworth-Heinemann, 1996.
- Hennermann JB., Gellermann J., Vollmer I, et al. Chronic kidney disease in adolescent and adult patients with phenylketonuria. J Inherit Metab Dis 2013;36;747.
- Lutsep HL, Albers GW, DeCrespigny A, et al. Clinical utility of diffusion-weighted magnetic resonance imaging in the assessment of ischemic stroke. Ann Neurol 1997;41:574-80.